Revealing How the Liver Regenerates After Injury
New findings reveal that immune signaling, not stem cells, drives liver repair.
Sponsored by

Sponsored by

A new study from Kyungpook National University challenges long-held beliefs about liver regeneration. Contrary to previous assumptions that stem cells drive repair, researchers discovered that a unique immune signaling pathway triggers liver cell growth after injury. These findings open exciting avenues for therapies that could enhance recovery in patients with liver damage, including those recovering from surgery or chronic disease. The research reveals a fresh perspective on how our liver heals itself naturally.
Image title: Harnessing Immune–Metabolic Pathways for Liver Repair
Image caption: Pericentral hepatocytes regulate liver regeneration through glutamate-mediated immune signaling.
Image credit: Dr. Eunjeong Kim from the Institute for Basic Science
License type: Original Content
Usage restrictions: Cannot be reused without permission
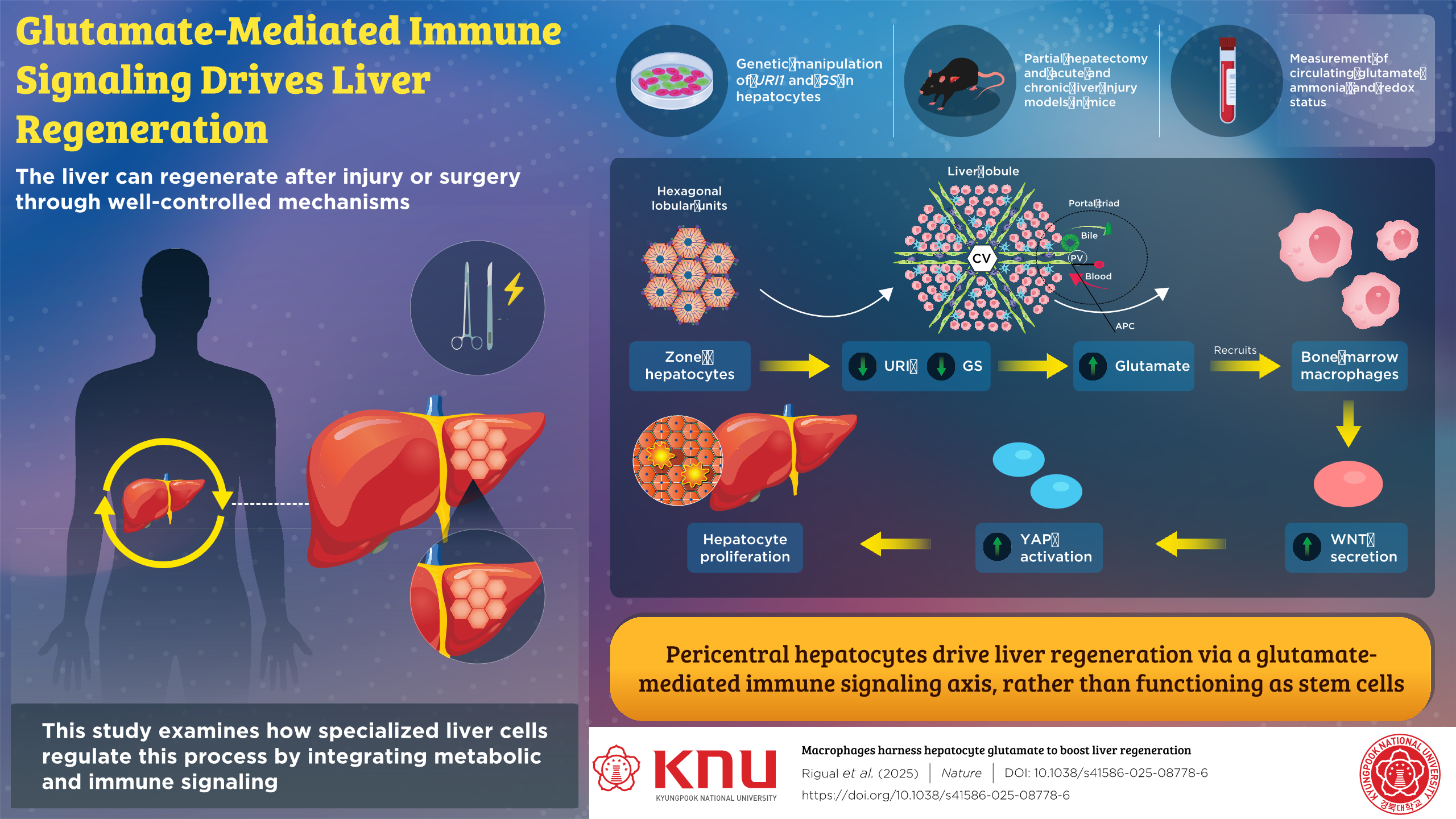
The liver has a remarkable capacity to regenerate itself following an injury or surgical removal, a property essential for maintaining metabolic homeostasis and survival after infection, toxin exposure, or partial hepatectomy. Unlike many organs that rely on resident stem cells, liver regeneration is driven largely by mature hepatocytes that re-enter the cell cycle. Despite decades of research, the mechanisms coordinating this response, particularly how metabolic cues integrate with immune signaling, have remained incompletely understood.
Pericentral (zone 3) hepatocytes were long proposed to function as a stem-like population responsible for liver regeneration. These cells are uniquely positioned near the central vein, where oxygen tension is low, and are defined by their expression of glutamine synthetase (GS), an enzyme that converts glutamate into glutamine. However, conflicting experimental evidence regarding the regenerative contributions of different hepatic zones has challenged this model, prompting the need for a more definitive mechanistic explanation. To address this issue, a research team led by Dr. Eunjeong Kim at Kyungpook National University set out to determine whether pericentral hepatocytes truly serve as stem cells or instead influence liver regeneration through alternative mechanisms. Their findings, published in Nature, on March 26, 2025, reveal a fundamentally different role for these cells.
Using genetically engineered mouse models, partial hepatectomy, and acute and chronic liver injury paradigms, the researchers systematically dissected pericentral hepatocyte function during regeneration. Central to their approach was hepatocyte-specific manipulation of URI1, a pericentral-enriched protein that regulates GS activity and glutamate metabolism. By modulating URI1 expression, the team controlled glutamate levels within the pericentral niche and assessed regenerative outcomes using biochemical assays, immune profiling, transcriptomics, and functional analyses.
The findings overturned the long-standing stem cell hypothesis. Pericentral hepatocytes did not act as a regenerative reservoir. Instead, they functioned as metabolic regulators that initiate liver regeneration through glutamate-mediated paracrine signaling. Loss of URI1 reduced GS activity, leading to glutamate accumulation locally and systemically. This metabolic shift reshaped the immune microenvironment of the regenerating liver. Elevated glutamate promoted the recruitment of bone-marrow–derived macrophages (BMDMs) to the pericentral zone.
Within macrophages, glutamate metabolism increased succinate levels, inhibiting prolyl hydroxylases and stabilizing the transcription factor HIF1α. Activated HIF1α directly induced Wnt3 expression. Macrophage-derived WNT3 then acted on hepatocytes to drive nuclear translocation of YAP1, a key regulator of cell proliferation and survival. Importantly, this regenerative pathway operated independently of canonical β-catenin signaling, revealing a previously unrecognized immune-driven WNT–YAP1 axis.
Beyond defining a new mechanism, the study demonstrated therapeutic potential. Dietary glutamate supplementation significantly enhanced liver regeneration across multiple disease models, including fibrosis and cirrhosis, without detectable toxicity at physiological doses. Analysis of human liver samples and public single-cell RNA-sequencing datasets revealed disruption of the URI1–GS–WNT3 axis in chronic liver disease, underscoring the translational relevance of the findings. Dr. Kim noted, “Nutritional glutamate supplementation can effectively promote liver regeneration and benefit patients with severe and chronic liver damage, such as those recovering after hepatectomy or even those awaiting a transplant.”
The implications extend beyond therapy. The study identifies a fundamental metabolic–immune circuit that coordinates liver repair and provides new targets for biomarkers and precision interventions. Dr. Kim emphasized, “Our results describe a fundamental and universal mechanism that allows the liver to regenerate after acute damage. These results may also help improve liver regenerative capacity in patients with severe liver damage, such as cirrhosis, or those who have undergone partial resection in surgery to remove a tumour.”
Together, these findings redefine pericentral hepatocytes as metabolic orchestrators of liver regeneration rather than stem cells, establishing a new framework for enhancing liver repair in acute and chronic disease.
Reference
Title of original paper
Macrophages harness hepatocyte glutamate to boost liver regeneration
Journal
Nature
DOI
About Kyungpook National University
Kyungpook National University (KNU) is a national university located in Daegu, South Korea. Founded in 1946, it is committed to becoming a leading global university based on its proud and lasting tradition of truth, pride, and service. As a comprehensive national university representing the regions of Daegu and Gyeongbuk Province, KNU has been striving to lead Korea’s national and international development by fostering talented graduates who can serve as global community leaders.
Website: https://en.knu.ac.kr/main/main.htm
About the author
Dr. Eunjeong Kim is an Assistant Professor of Department of Biology and invited Research fellow Center for Genome Engineering Research at IBS (Institute for Basic Science). Her group is developing approaches to study liver diseases such as hepatocellular carcinoma and cirrhosis etc. Kim’s group is also interested in cancer research including head and neck squamous cell carcinoma. Her group is trying to understand how diseases were developed and which mechanisms were existed. Before coming to Kyungpook National University, she completed the Postdoctoral training at Nabil Djouder’s group at CNIO (Centro Nacional de Investigaciones Oncológicas) in Spain.