Wei Xia's Team | "Disordering" Crystals Leads to Unexpected Low-Cost Solid-State Electrolytes
Scientists developed an amorphous halide electrolyte with lower lithium content, high ionic conductivity, strong air stability, and long battery cycling performance.

Sponsored by

Sponsored by

In the realm of materials science, well-ordered crystalline structures have long been regarded as the high achievers. This conventional mindset has led to the entrenched belief that halide electrolytes must be lithium-rich to be highly conductive and crystalline to be effective. Consequently, the exploration of ion conduction in amorphous materials has been relatively limited for an extended period.
Wei Xia, Assistant Professor at the Eastern Institute of Technology, Ningbo (EIT), and his collaborators have taken a different approach—by disordering crystals, they yielded an unexpected outcome. They successfully synthesized a novel class of amorphous (i.e., lacking a regular crystalline structure) halide electrolytes. This seemingly chaotic material not only significantly reduces dependence on scarce lithium resources but also exhibits surprising robustness, offering a new pathway to overcome the commercialization hurdles of all-solid-state batteries.
Their findings were published in Nature Communications on March 1st (Beijing time). The Eastern Institute of Technology, Ningbo, is the primary affiliation of the paper.
Breaking the "Ceiling" of Crystals: Amorphous Materials Step Up
Within the solid-state battery family, halide electrolytes have always been star players—they conduct ions rapidly and are compatible with high-voltage cathodes. However, a significant drawback is that to achieve high ionic conductivity, a substantial amount of lithium is required, typically constituting over 4.3% of the total material mass.
Lithium itself has limited global reserves, is expensive, and is particularly hygroscopic, readily absorbing moisture from the air and degrading. This necessitates the production and storage of solid-state batteries in extremely dry environments, keeping costs prohibitively high. Many experts in the field also believe that traditional crystalline materials have hit a performance ceiling—making them significantly cheaper or more stable has become exceedingly difficult.
The research team decided to challenge the paradigm: since the crystalline structure constrains our imagination, why not abandon crystals altogether?
Using a simple mechanical ball milling method, they successfully synthesized a series of amorphous halide electrolytes, designated as xLi₂SO₄-ZrCl₄. In simple terms, it means the internal atomic arrangement lacks the long-range order of crystals. It is precisely this disorder that yielded unexpected advantages:
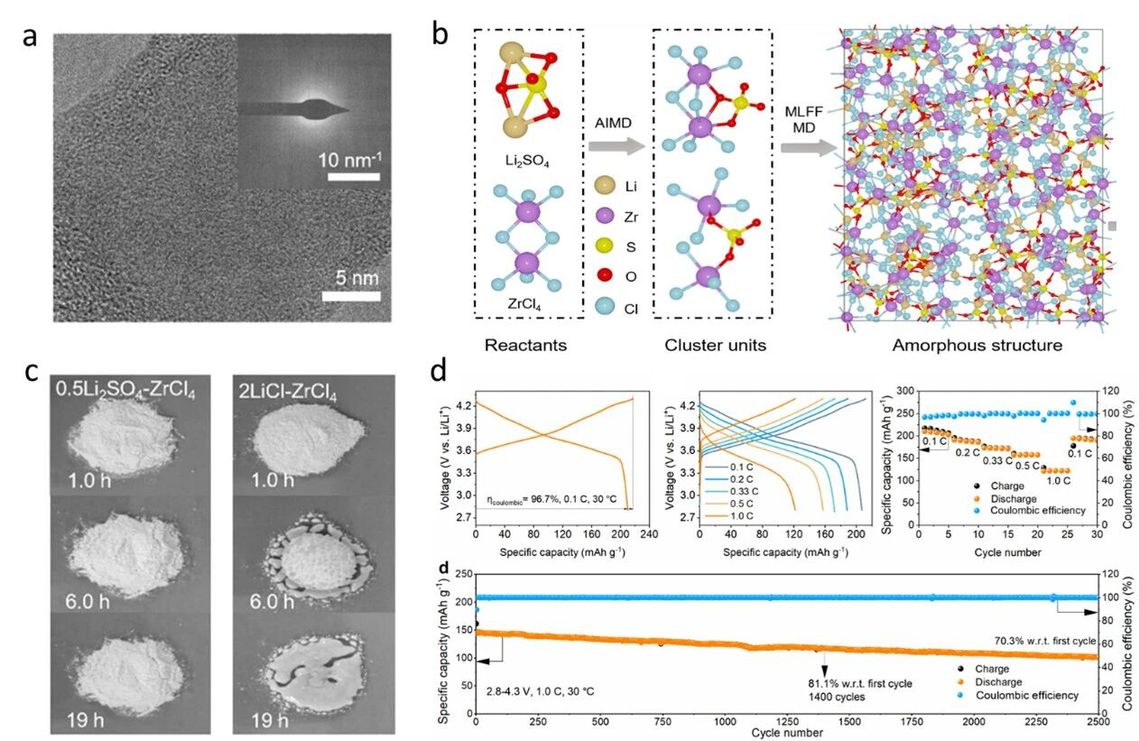
Structure and properties of 0.5Li2SO4-ZrCl4. Image provided by the research group
A "Leaner" Lithium Composition:
By weight, the optimal composition, 0.5Li₂SO₄-ZrCl₄, contains only 2.4% lithium, significantly lower than the 5-7% typically required for conventional crystalline materials. Crucially, this reduction does not compromise ionic conductivity—it exhibits a room-temperature ionic conductivity of 1.5 mS cm⁻¹, comparable to state-of-the-art crystalline halide electrolytes.
Building "Highways" for Lithium Ions:
Leveraging advanced characterization techniques and computational simulations, the team elucidated, for the first time, the conduction mechanism within this amorphous material. They discovered that the material forms a disordered framework network composed of SO₄²⁻ anions and Zr-Cl structural units. This network effectively eliminates the energy barrier heterogeneities present in crystalline materials, allowing lithium ions to hop rapidly and, consequently, migrate more smoothly.
Unexpected "Toughness":
The most significant vulnerability of conventional crystalline halide electrolytes is their sensitivity to air. However, this amorphous material demonstrated exceptional air stability. Even when exposed to a relative humidity of 30%, its performance degradation and moisture uptake rates were substantially lower than those of comparable crystalline materials. This suggests that, in a future production line, the prohibitively expensive absolutely dry manufacturing environments might no longer be necessary, significantly lowering the entry barrier for production.
Enduring 2500 Cycles: A "China Solution" for Future Batteries
To validate the material's real-world applicability, the team assembled and tested all-solid-state lithium batteries.
The results were compelling. Under standard testing conditions, the initial discharge capacity reached 209.9 mAh g⁻¹, approaching the theoretical value. At a 1.0 C rate, the battery retained over 81% of its capacity after 1400 cycles. Remarkably, after 2500 cycles, it still delivered a discharge capacity of 101.7 mAh g⁻¹.
Furthermore, the battery demonstrated commendable stability even with thicker cathode coatings or under higher operating voltages.
The innovation of this research lies in the pioneering introduction of anion clusters into the design of halide solid-state electrolytes, challenging the long-held notion that high lithium content is necessary for high ionic conductivity in this material class. Through chemical design, the team achieved performance on par with traditional materials while simultaneously reducing cost and enhancing stability. This novel electrolyte offers a viable China’s Solution for the commercialization of all-solid-state lithium batteries. It brings the prospect of safer, more affordable, and longer-range electric vehicles a significant step closer.
Wen Tang, Ph.D. Student; Feilong Wang, Postdoctoral Researcher; and Shuaika Liang, Ph.D. Student from EIT are the co-first authors of the paper. Assistant Professor Xia Wei; Assistant Professor Shuo Wang; Chair Professor Xueliang Sun, Fellow of the Canadian Academy of Engineering and Foreign Member of the Chinese Academy of Engineering from EIT, and Chair Professor Zifeng Ma from Shanghai Jiao Tong University are corresponding authors.
This research was supported by the National Natural Science Foundation of China, the Zhejiang Provincial Natural Science Foundation of China, the Natural Science Foundation of Ningbo, and the China Postdoctoral Science Foundation. The work also benefited from testing support at major scientific facilities, including the China Spallation Neutron Source (CSNS), the Shanghai Synchrotron Radiation Facility (SSRF), and the SPring-8 synchrotron in Japan.