Strategy for Valorizing Methane
Zhangxing Chen and Heng Zhao (EIT), with Yujie Xiong, developed a stable catalyst for efficient methane and CO₂ conversion under mild conditions.
Sponsored by

Sponsored by

The greenhouse gases methane and carbon dioxide are major contributors to global warming. Dry reforming of methane (DRM, CH₄ + CO₂→ 2CO + 2H₂) is a process capable of converting both gases simultaneously into useful syngas. However, this reaction requires temperatures above 700°C, and catalysts are prone to rapid deactivation, primarily through coking, which has hindered its large-scale industrial adoption.
A significant advancement has now been made by the team led by Zhangxing Chen, Chair Professor and Foreign Member of the Chinese Academy of Engineering, and the team led by Assistant Professor Heng Zhao, both at the Eastern Institute of Technology, Ningbo (EIT), in collaboration with Professor Yujie Xiong from the Southern University of Science and Technology. They have designed a novel catalyst that not only dramatically improves reaction efficiency under milder conditions but also maintains long-term operational stability, paving a new way for the efficient resource utilization of methane and carbon dioxide.
The related research has been published in Nature Communications, with the Eastern Institute of Technology, Ningbo listed as the primary affiliation.
Bypassing Lattice Oxygen: A New Catalyst Achieving Record Efficiency and Stability
Conventional DRM relies heavily on metal oxide catalysts. These catalysts utilize reactive lattice oxygen stored within their structure to participate in the reaction and remove carbon deposits. However, lattice oxygen possesses poor mobility; it is consumed rapidly but replenished slowly. Its excessive depletion leads to structural collapse of the catalyst and aggregation of metal nanoparticles, resulting in rapid deactivation.
The research team identified this fundamental limitation and proposed an innovative strategy: bypassing the need for lattice oxygen by directly leveraging reactive oxygen species generated in situ during the reaction.
They successfully designed and synthesized a novel catalyst termed N-Ni/NiCo@C. Its key innovation lies in the absence of traditional metal oxide lattice oxygen. Instead, the catalyst utilizes incoming CO₂ molecules, which dissociate on its surface to produce highly active oxygen atoms or hydroxyl radicals in real time. These in situ generated active oxygen species participate more directly and efficiently in methane activation and conversion while simultaneously removing carbon deposits as they form, thereby preventing deactivation caused by lattice oxygen exhaustion.
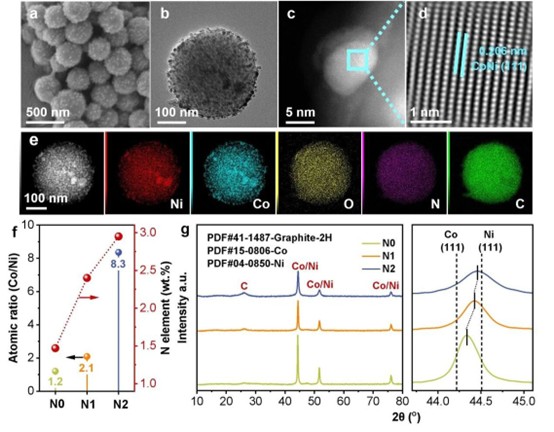
Morphology and structural characterization of the catalyst. Image provided by the research group.
Experimental results demonstrate the outstanding performance of this new catalyst, which exhibits both high efficiency and remarkable stability.
Under relatively mild conditions of 540°C with light irradiation, the catalyst achieved a high photo-to-chemical energy conversion efficiency of 52%. This indicates its ability to convert over half of the input photon energy into chemical energy stored within the syngas, a performance surpassing most previously reported catalysts.
In a durability test spanning 200 hours of continuous operation, the catalyst maintained its activity without significant decline. In contrast, conventional catalysts deactivate rapidly under similar conditions due to coking and sintering, underscoring the superior stability of the new design.
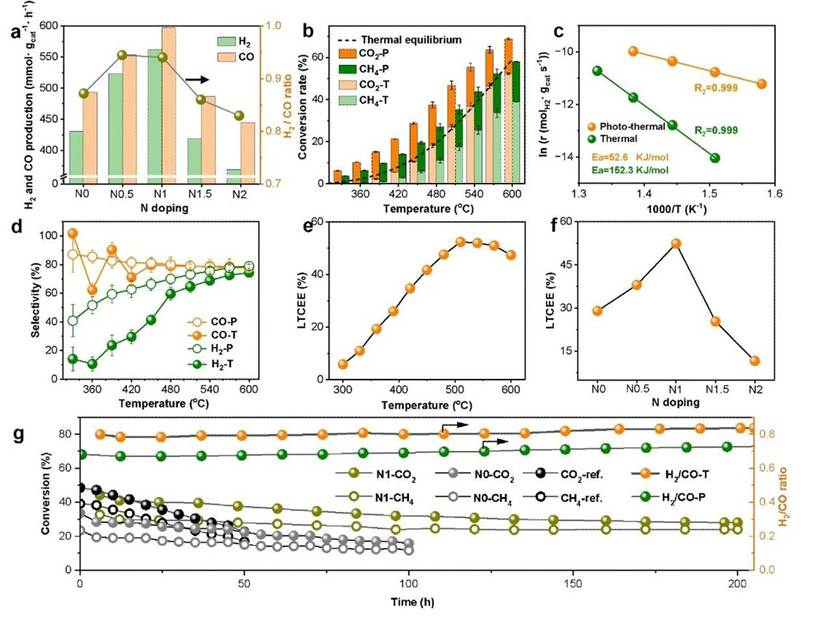 Excellent photothermal DRM catalytic performance. Image provided by the research group.
Excellent photothermal DRM catalytic performance. Image provided by the research group.
A Directed Electron Channel Enabling Highly Efficient Methane Conversion
What underpins this high efficiency and stability? Through advanced characterization, the team elucidated the mechanism. The catalyst comprises uniformly dispersed nickel-cobalt alloy nanoparticles encapsulated within a nitrogen-doped graphitic carbon shell.
The critical feature resides in this carbon shell and the doped nitrogen atoms.
The nitrogen atoms form a unique C–N–Ni coordination structure with the carbon layer and nickel atoms. This structure acts as a directed electron channel, precisely and rapidly channeling photogenerated electrons to the NiCo active sites. This process prevents oxidation and deactivation of the metal sites.
Furthermore, this electron channel modulates the surface electronic distribution of the alloy, inducing a natural functional differentiation: nickel sites show a higher propensity for activating methane, while cobalt sites are more favorable for activating carbon dioxide. This efficient synergy significantly boosts the overall reaction rate.
The study also revealed that light irradiation does not merely provide thermal energy but opens a distinct reaction pathway. Under light, activated hydrogen atoms react with CO₂ to form a key carboxyl intermediate. The decomposition of this intermediate yields active hydroxyl species, which function like cleaners to promptly oxidize carbon-forming intermediates, enabling a self-cleaning mechanism during operation.
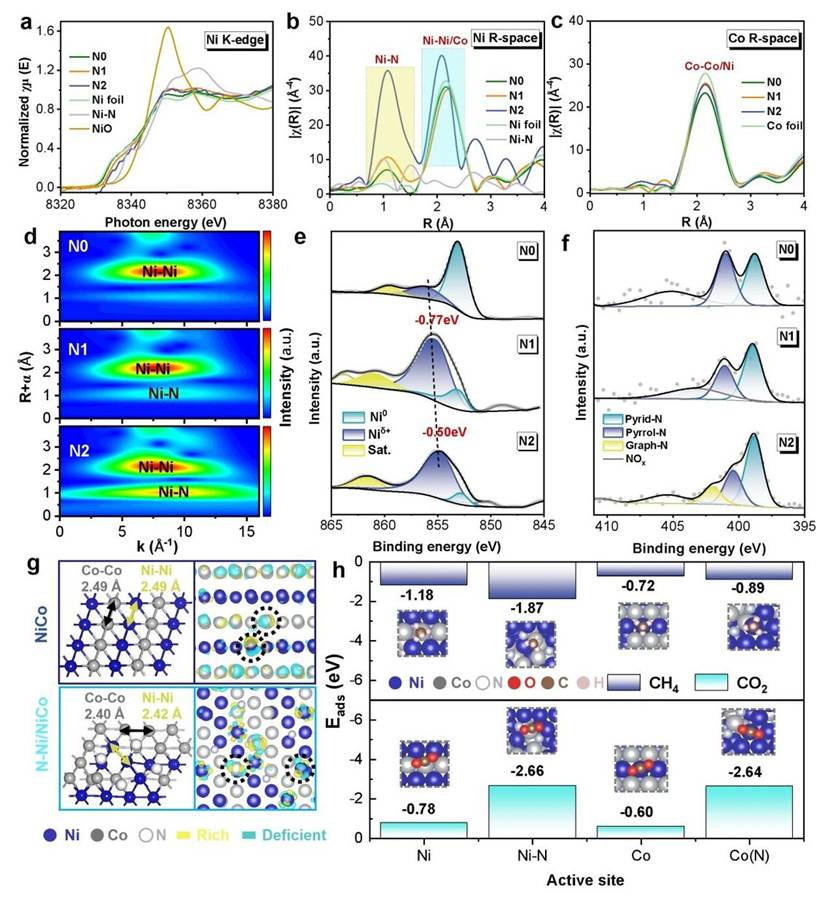 Analysis of the atomic coordination environment and electronic structure. Image provided by the research group.
Analysis of the atomic coordination environment and electronic structure. Image provided by the research group.
This work reports the first high-loading (30 wt%), lattice-oxygen-free NiCo alloy-based photothermal catalyst for DRM. By constructing the C–N–Ni coordination for directed electron transfer, it successfully resolves the traditional conflict between activity (sintering) and stability. The research demonstrates that utilizing in situ generated surface active oxygen species (O*/OH*) to directly oxidize reaction intermediates is a viable strategy to supplant the conventional lattice oxygen mechanism. This breakthrough provides a new perspective for designing efficient and stable photothermal catalytic systems and is anticipated to have a significant impact on the fields of catalysis, energy science, and carbon neutrality efforts.
Tingting Pan, Postdoctoral researcher at EIT and Weiwei Xu PhD candidate at the University of Liverpool are co-first authors of the paper. Zhangxing Chen, Heng Zhao, and Yujie Xiong are the corresponding authors. Collaborators also include the team of Researcher Hua Deng from the Institute of Urban Environment, Chinese Academy of Sciences.
This project received support from the National Natural Science Foundation of China, the China Postdoctoral Science Foundation, the Young Innovative Talent of Yongjiang Talent Project, theNingbo City Natural Science Foundation, and others.