Cancer Metastasis: Reducing "Special Fuel" Supply
A study led by Prof. Jun Yu and Prof. Zijun Huang (CUHK) with Prof. Zongwei Cai (EIT Ningbo) reveals BCAA metabolism drives colorectal cancer metastasis via the UMP–Vimentin axis.
Sponsored by

Sponsored by

A collaborative study led by Professor Jun Yu and Professor Zijun Huang from The Chinese University of Hong Kong, together with Chair Professor Zongwei Cai from the Eastern Institute of Technology, Ningbo, has demonstrated that branched-chain amino acid (BCAA) metabolism drives colorectal cancer metastasis through the uridine monophosphate-vimentin (UMP-Vimentin) axis.
This work is the first to establish that the expression levels of the cytosolic branched-chain amino acid transaminase (BCAT1) and its mitochondrial isoform (BCAT2) can predict metastatic risk in colorectal cancer patients—a finding potentially applicable to multiple cancer types.
The related findings were recently published in Cell Metabolism.
Colorectal cancer metastasis is a highly complex process involving profound metabolic reprogramming. Using metabolomic screening, the researchers identified significant dysregulation of BCAA metabolism during metastatic progression. Delving into the downstream enzymes of BCAA catabolism, they found that expression of the cytosolic enzyme BCAT1 was upregulated, whereas expression of the mitochondrial isozyme BCAT2 was downregulated.
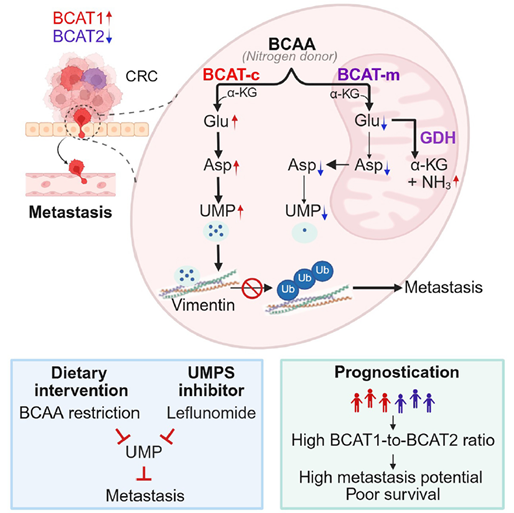
Compartmentalized BCAA metabolism in the cytosol versus mitochondria regulates colorectal cancer metastasis. Image provided by the research team.
Through analysis of clinical cohorts, the research team elucidated the predictive significance of cytosolic versus mitochondrial BCAA metabolism for cancer metastasis. They validated the pro-metastatic function of the transaminase BCAT1 and the anti-metastatic function of the isozyme BCAT2 in both cellular and animal models of metastasis. By genetically swapping the subcellular localizations of BCAT1 and BCAT2, the team demonstrated that the subcellular compartmentalization of BCAT enzymatic activity is critically important. Finally, employing metabolomics and ¹⁵N/¹³C isotope tracing experiments, they uncovered the underlying mechanism: BCAT1 and BCAT2 differentially regulate cellular uridine monophosphate (UMP) levels, which stabilizes vimentin by protecting it from ubiquitin-mediated degradation, thereby driving cell migration.
The findings demonstrate that during in vivo metastasis, colorectal cancer cells utilize dietary branched-chain amino acids as a special fuel to acquire migratory and invasive capabilities. Therefore, devising and implementing precise nutritional interventions to restrict the supply of this special fuel represents a novel strategy to inhibit cancer cell dissemination.
Moreover, the researchers identified that a critical node within this fuel supply pathway can be effectively targeteds by Leflunomide, a drug commonly used to treat rheumatoid arthritis. This discovery opens a promising and expedited avenue for combating colorectal cancer metastasis through drug repositioning—repurposing an existing therapeutic into a novel agent against cancer spread. This approach promises to significantly shorten the translational timeline from laboratory discovery to clinical application, offering new avenues for the personalized management of patients with advanced colorectal cancer.
The first author of the paper is Dr. Fenfen Ji, a postdoctoral research fellow at The Chinese University of Hong Kong. The corresponding authors are Professor Zongwei Cai, Professor Jun Yu, and Professor. Zijun Huang.